
Medical Device Regulations Extended By EU
IVDR/MDR Regulations Extended to Combat Medical Device Supply Shortage Recently, a significant development has come to pass concerning the implementation of the EU’s new medical device legislation.
Read the latest updates on the medical device business development industry as well as company updates, the latest case-studies and whitepapers.


IVDR/MDR Regulations Extended to Combat Medical Device Supply Shortage Recently, a significant development has come to pass concerning the implementation of the EU’s new medical device legislation.

Per April 2023 Canadian medical manufacturers can apply for a funding for fiscal year 2023 and 2024! Don’t miss out on this opportunity and read how to

If you are looking to expand your medical device sales in Europe, dont miss out on this complete list! Tradeshows remain a big source for customer meetings

Accessing the Swiss medical device market with FDA approval The swiss medtech approves medical device manufacturers enter the Swiss healthcare market with FDA. This is good news
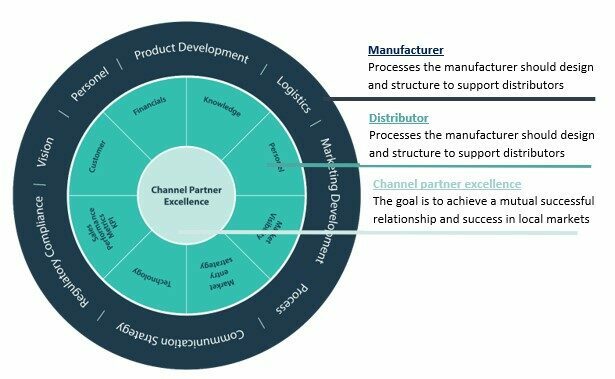
Optimizing your international medical device distributors You may or may not have had your annual sales meetings with your distributors, if you haven’t it’s a critical

Do not miss an opportunity to meet us at Arab Health exhibition in Dubai from 30th January to 2nd February. GrowthMedics Team will be attending this year’s

International Expansion Strategies International expansion strategies for medical companies. Expanding Into new markets can be challenging and complex. Evaluating expansion strategies from your strength and assessing market

If you are attending or exhibiting at Medica 2022 🤝 Let’s meet up to explore your Global Growth and Importing opportunities in Europe and the Middle East!

Tell us how we can help your business succeed in Europe. We have expertise that can help your business to become a top manufacturer in Europe. We will send you an opportunity that can help you grow your business.