Starting your EUDAMED registration is no longer optional for MedTech companies aiming to meet the mandatory May 2026 requirements.
The complexities of European medical device regulations can feel like a labyrinth. For MedTech CEOs and Quality Managers, understanding and mastering the European Database on Medical Devices (EUDAMED) isn’t just a regulatory checkbox; it’s a critical strategic imperative for EU market access and maintaining post-market surveillance integrity.
At Growth Medics, we understand the immense pressure you face. This comprehensive guide cuts through the noise, offering clarity on EUDAMED registration and how proactive engagement can de-risk your operations and accelerate your growth trajectory in the European and Middle Eastern markets.
What is EUDAMED and Why is it Indispensable for Your Business?
EUDAMED is the central IT system developed by the European Commission to implement the Medical Devices Regulation (MDR (EU) 2017/745) and the In Vitro Diagnostic Regulation (IVDR (EU) 2017/746). It serves as a vital tool for:
- Enhanced Transparency: Providing public access to key information on medical devices.
- Improved Traceability: Tracking devices throughout their lifecycle from manufacturing to end-use.
- Strengthened Post-Market Surveillance: Enabling swift identification and response to safety issues.
- Coordinated Market Surveillance: Facilitating cooperation among National Competent Authorities.
While its full mandatory implementation has seen delays, the operational modules are crucial for any economic operator aiming for compliant and sustainable presence in the EU market. Ignoring EUDAMED today means risking significant disruption tomorrow.
Current Status and Strategic Importance of Voluntary EUDAMED Engagement
As of now, EUDAMED operates on a voluntary basis for several key modules, with the projected date for full mandatory compliance often adjusted based on system readiness. The current consensus points towards a mandatory application 24 months after all six modules are officially declared fully functional.
However, viewing this as a reason to delay is a misstep. Proactive, voluntary registration in the available modules offers significant advantages:
- Pre-emptive Compliance: Familiarity with the system ensures a smoother transition when mandatory deadlines arrive.
- Operational Efficiency: Early data entry reduces last-minute rushes and potential data quality issues.
- Enhanced Credibility: Demonstrates your commitment to regulatory excellence to Notified Bodies, Competent Authorities, and partners.
- Market Access Catalyst: Certain stakeholders, including some distributors and healthcare providers, may increasingly prefer working with manufacturers already registered in EUDAMED.
Decoding the EUDAMED Modules: A Strategic Overview
EUDAMED is structured into six interconnected modules, each serving a distinct purpose within the regulatory framework. Understanding them is key to your compliance strategy.
1. Actor Registration Module (Voluntarily Available Since Dec 1, 2020)
This module is your entry point into EUDAMED. All economic operators—manufacturers, Authorized Representatives (ARs), importers, and system/procedure pack producers—must register to obtain a unique Single Registration Number (SRN). The SRN is your digital identity within EUDAMED and is indispensable for all subsequent interactions.
Key Steps for Actor Registration:
- Role Selection: Identify your specific economic operator role (e.g., manufacturer, Authorized Representative).
- Organization Details: Provide comprehensive information about your legal entity.
- Declaration on Information: Sign and upload the required declaration confirming data accuracy.
- Competent Authority Verification: Your application is reviewed by the relevant National Competent Authority.
Growth Medics’ Insight: For non-EU manufacturers, your EU Authorized Representative (EC-REP) plays a critical role in verifying your actor registration request before it reaches the National Competent Authority. Growth Medics acts as your trusted Authorized Representative, streamlining this process and ensuring your registration is accurate and compliant, thereby securing your SRN efficiently.
2. UDI Database and Registration of Devices Module (Voluntarily Available)
The Unique Device Identification (UDI) system is a cornerstone of traceability under the MDR/IVDR. This module is where you register information about your devices, including their UDI-DI (Device Identifier).
MDR Article 27 mandates that devices, and certain aspects of their packaging, carry a UDI. The UDI database acts as the central repository for this critical information, enabling rapid identification and tracing of specific devices on the market.
Key Considerations for UDI Registration:
- UDI Issuing Agencies: You must obtain your UDI from one of the European Commission-designated issuing agencies (e.g., GS1, HIBCC, ICCBBA, IFA).
- Data Accuracy: Precise and consistent data entry is paramount, as this information will be publicly accessible for most devices.
- Life-Cycle Management: The UDI data must be maintained and updated throughout the device’s lifecycle.
Growth Medics’ Insight: Compiling and submitting accurate UDI data can be resource-intensive. Our Regulatory Affairs specialists can assist in collating necessary information, liaising with UDI issuing agencies, and ensuring your device data meets EUDAMED’s stringent requirements, reducing your internal burden and compliance risk.
3. Notified Bodies and Certificates Module (Voluntarily Available)
This module focuses on the crucial role of Notified Bodies in MDR/IVDR compliance. It centralizes information regarding:
- Notified Body Designations: Details of organizations authorized to assess conformity.
- Certificates: All certificates issued for devices, including initial certifications, amendments, suspensions, reinstatements, and withdrawals.
Key Impact: This module provides transparency on the conformity assessment process, allowing stakeholders to verify the validity of device certifications and the scope of Notified Body approvals.
4. Clinical Investigations and Performance Studies Module (Not Yet Fully Available)
Anticipated to be fully operational soon, this module will create a unified system for managing clinical investigations for medical devices and performance studies for in vitro diagnostic devices. It aims to streamline the application, assessment, and reporting processes, fostering greater transparency and collaboration.
5. Vigilance and Post-Market Surveillance Module (Not Yet Fully Available)
This module is critical for enhancing device safety. It will facilitate the reporting and analysis of serious incidents, Field Safety Corrective Actions (FSCA), and trend reports. Its full activation will significantly improve the post-market surveillance (PMS) activities required under MDR/IVDR, enabling quicker responses to safety concerns across the EU.
6. Market Surveillance Module (Not Yet Fully Available)
This module is designed to empower National Competent Authorities in their market surveillance activities. It will serve as a platform for sharing information on non-compliant devices, enforcement actions, and measures taken to withdraw unsafe devices from the market. This module will enhance the ability of authorities to protect public health more effectively.
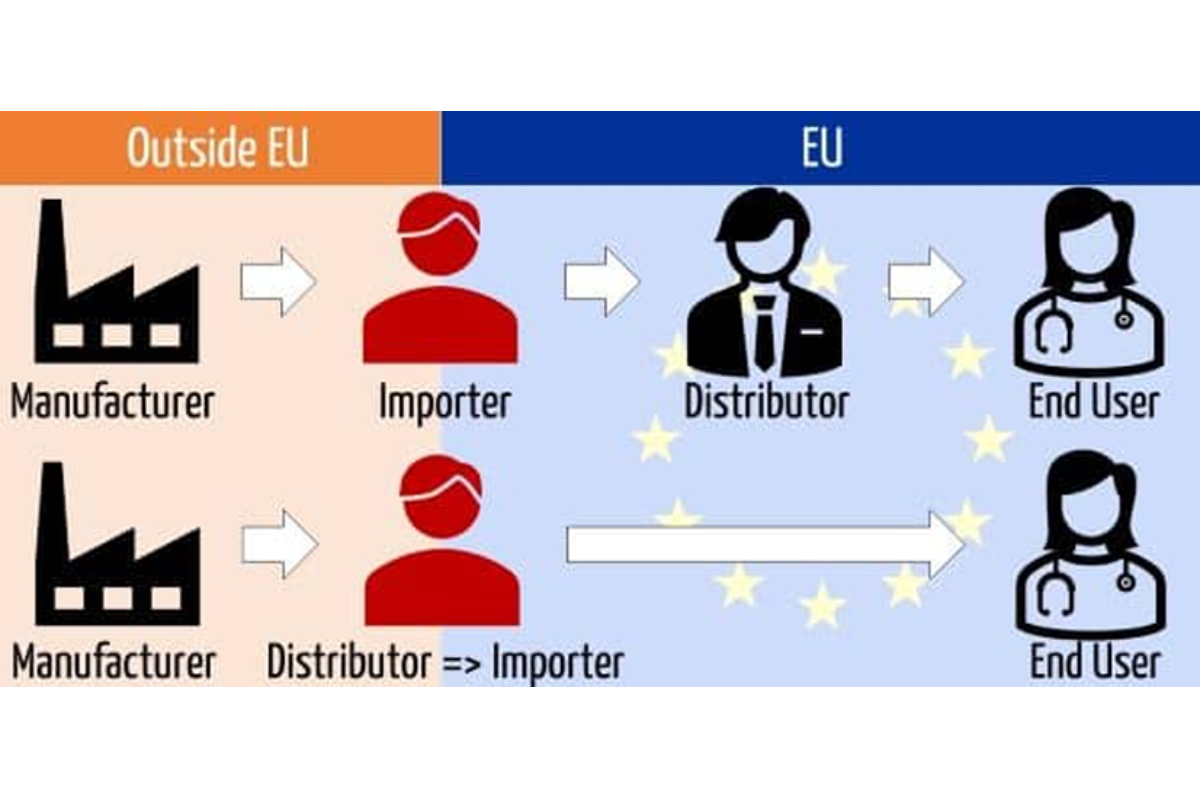
The Critical Role of Importer Registration
Beyond manufacturers and Authorized Representatives, importers (as defined by MDR Article 13) also hold significant responsibilities. It is strongly recommended that your EU importer is officially registered with the National Competent Authority and possesses their own SRN.
With an SRN, the importer can officially link themselves to the specific manufacturer and product groups they are responsible for within EUDAMED. This ensures a clear chain of responsibility, traceability, and accountability throughout the supply chain, which is essential for rapid response in case of vigilance events or market surveillance actions.
Strategic Recommendations for MedTech Leaders
Navigating EUDAMED and the broader MDR/IVDR landscape requires foresight and robust execution. Here are five actionable insights to ensure your organization maintains compliance and fosters sustainable growth:
- Proactively Secure Your SRN and Engage Voluntarily: Do not wait for mandatory deadlines. Initiate Actor Registration immediately. Securing your SRN early establishes your presence and allows you to gain familiarity with EUDAMED, mitigating future compliance risks.
- Leverage Your EU Authorized Representative for EUDAMED Management: For non-EU manufacturers, your EU Authorized Representative is not just a legal contact; they are your strategic partner in EUDAMED. Ensure they are capable of verifying your actor registration, supporting UDI data submission, and helping manage your EUDAMED presence. Growth Medics provides comprehensive Authorized Representation services specifically designed for this purpose.
- Invest in Robust Data Management and Quality: The accuracy and completeness of your data within EUDAMED are paramount. Implement internal processes and systems to ensure high-quality data input for UDI, device details, and all other required information. Poor data quality can lead to delays, non-compliance, and operational inefficiencies.
- Develop a Comprehensive EUDAMED Readiness Strategy: Integrate EUDAMED compliance into your overall Quality Management System (QMS). This includes defining roles and responsibilities, establishing training programs for relevant personnel, and setting up internal procedures for data submission and maintenance across all relevant modules.
- Partner with Expert Regulatory Affairs Consultants: The nuances of EUDAMED are complex and constantly evolving. Engaging with seasoned Regulatory Affairs professionals, like Growth Medics, provides access to up-to-date expertise, streamlines your registration processes, and ensures your long-term market access strategy is sound. We don’t just guide you; we actively manage these complexities for you.
At GrowthMedics, we are more than consultants; we are your strategic partner in achieving seamless EUDAMED compliance and accelerating your medical device market entry in Europe and the Middle East. From Authorized Representation to comprehensive Regulatory Affairs support, we’re here to transform regulatory challenges into growth opportunities.
Contact GrowthMedics today to discuss your EUDAMED strategy and ensure your path to European market success.