The implementation of EU MDR and IVDR is a journey of critical milestones, and the most significant one is now upon us. As the mandatory transition to EUDAMED approaches, your strategy for Medical Device EU Importing 2026 must evolve to maintain market access.
For non-EU manufacturers, holding a CE mark is no longer the final step. To maintain market access, you must now navigate the phased implementation of EUDAMED, the European database for medical devices. This isn’t just a digital filing cabinet; it is the backbone of transparency and safety in the EU.
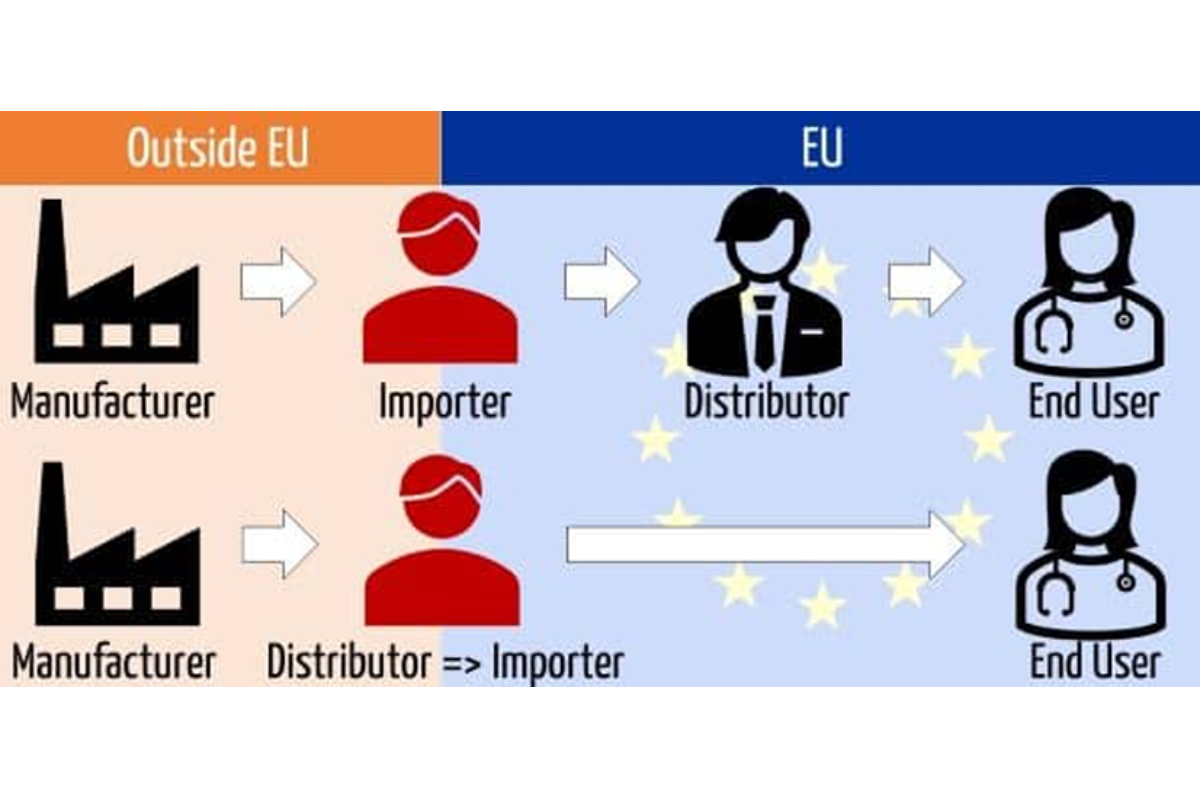
Now is the moment to audit your importing strategy. As EUDAMED modules for Actor and Device registration become mandatory, your choice of Importer determines your ability to generate a Single Registration Number (SRN) and keep your supply chain moving.
Why the “Distributor-as-Importer” Model can create bottlenecks
With stricter EUDAMED oversight and Notified Body bottlenecks, the old way of importing can become a liability:
- Commercial “Lock-in” & Market Conflict: Tying your legal importer address to one distributor makes switching partners or adding new sales channels a labeling nightmare. You lose the flexibility to scale a multi-channel strategy.
- The Liability & Capability Gap: Importers now share joint liability for compliance. Most distributors lack the RA/QA infrastructure to manage EUDAMED Actor profiles or perform mandatory Article 13 checks.
- Administrative Burnout: EUDAMED requires importers to link themselves to your devices and update data within one week of any change. Many distributors are now refusing these burdens, stalling your product launch.
What You’ll Discover in This Practical Guide:
- The 2026 Compliance Roadmap: Key deadlines for Actor and Device registration in EUDAMED.
- Regulatory vs. Fiscal Silos: How to design a setup where compliance and cash flow (VAT Article 23) work in harmony.
- The Netherlands Advantage: Why the Dutch “Hub” is the trending choice for centralized EU medical device logistics.
- The “Importer Onboarding Pack”: A checklist of exactly what you need to prepare—from UDI-DI mapping to Quality Agreements—to avoid shipment holds.
Ready to guarantee importer compliance, place your medical device and In Vitro Devices seamless, efficient and independent in the EU, UK and CH markets?
GrowthMedics provides the independent, ISO 13485-standard importing infrastructure you need to bypass distributor conflict and EUDAMED delays.
- Total Flexibility: Add or change distributors without relabeling.
- Data Security: Your technical documentation stays with an independent regulatory partner, not a sales agent.
- Fiscal Efficiency: Leverage our Netherlands-based expertise for seamless customs and VAT management.