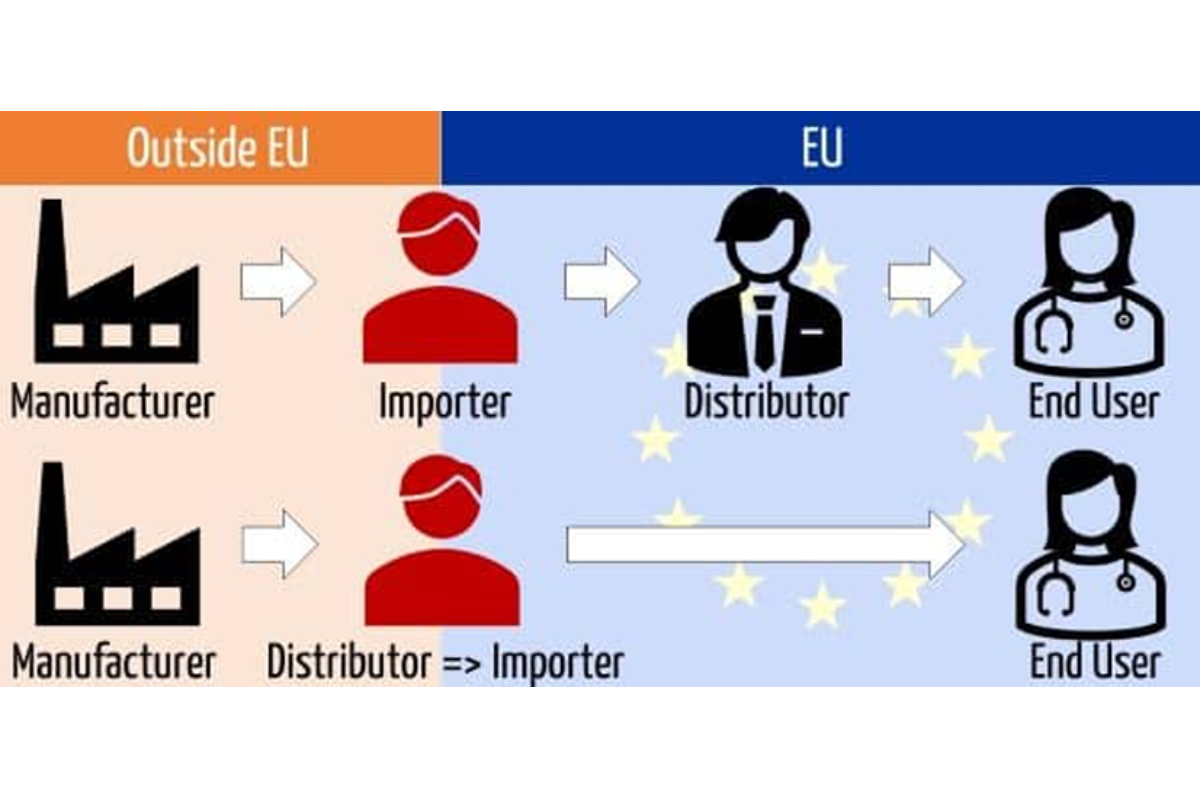
European veterinary market insights
At GrowthMedics, we are proud to support global veterinary device manufacturers in establishing a robust presence in the dynamic European veterinary healthcare market. By leveraging our expertise in market development, we offer unparalleled sales support to facilitate seamless market entry and sustained growth in Europe and the MENA regions. From navigating complex regulatory landscapes to tailoring sales strategies for diverse markets, we serve as a strategic ally. With access to an extensive network, industry insights, and dedicated sales professionals, we empower veterinary manufacturers to excel in the ever-evolving veterinary market. European Veterinary Market size and Projections Market Valuation in 2024 The European veterinary healthcare market is valued at an impressive USD 11.4 billion, reflecting its critical economic importance within the region. Growth Projections (2024–2028) Compound Annual Growth Rate (CAGR): 6.2% Projected Market Value by 2028: USD 14.7 billion This steady growth underscores the resilience and rising demand for veterinary products and services in Europe. Key Trends in the European Veterinary Market Rise of Telemedicine in Veterinary Healthcare The adoption of telemedicine is revolutionizing pet care in Europe. Remote consultations are becoming a preferred option for pet owners, enabling convenient access to veterinary services. The European veterinary telemedicine market is projected to grow at a CAGR of 18% from 2023 to 2028. Integration of Advanced Diagnostics Technologies such as molecular diagnostics and imaging are transforming the veterinary diagnostics market, which is expected to surpass USD 1.2 billion by 2024. These advancements are enhancing the precision and speed of diagnoses in veterinary practices. Focus on Animal Welfare Legislation The European Union’s stringent animal welfare laws are driving higher standards for healthcare practices. These regulations ensure ethical treatment, further boosting demand for innovative solutions in veterinary healthcare. Growth of Veterinary Pharmaceuticals With the increasing prevalence of chronic diseases in animals, the demand for veterinary pharmaceuticals continues to rise. The market is valued at USD 5.7 billion in 2024, with a CAGR of 5.8%. Increased Pet Insurance Adoption Pet insurance adoption is growing rapidly, with penetration rates reaching 25% in Europe. The Nordics lead this trend at ~30%, followed by the UK at ~20%. This growth is fueling investment in veterinary healthcare services. Veterinary OEM Markets in Europe The European veterinary OEM market is diverse, with several countries playing key roles in manufacturing veterinary devices and equipment. Germany: Leading the Veterinary Market Germany accounts for the largest share of the OEM market, valued at 4.5 billion Euros in 2024. Known for its precision engineering and innovative technologies, Germany is at the forefront of veterinary device production. The United Kingdom The UK excels in research and innovation, supplying high-quality veterinary devices across Europe. France, Netherlands, and Spain France specializes in diagnostic imaging and surgical equipment, while the Netherlands has emerged as a strategic hub for manufacturing. Spain contributes significantly to advancing veterinary solutions tailored to evolving market demands. Innovative Startups in the Veterinary Sector Here are some companies revolutionizing the European veterinary market in 2024: Felmo: Felmo is a Berlin-based startup that is transforming the way pet owners access veterinary care. The company offers a mobile app that connects pet owners with veterinarians for at-home consultations, vaccinations, and other services. Felmo’s platform is designed to make veterinary care more convenient and accessible for pet owners, particularly those who live in rural areas or have difficulty taking their pets to traditional veterinary clinics. Dalma: Dalma is a Paris based start-up which is a pet health monitoring platform that uses artificial intelligence (AI) to identify potential health issues in pets. The company’s platform uses a wearable device to track a pet’s activity, sleep patterns, heart rate, and other vital signs. Dalma’s AI algorithms analyze this data to identify patterns that may indicate a developing illness. Lassie: Lassie is a Swedish fintech startup that is revolutionizing pet insurance with a personalized approach that focuses on prevention. The company’s platform uses a combination of data science, AI, and machine learning to assess a pet’s risk factors and provide personalized recommendations for insurance coverage and preventative care. Upcoming Veterinary Market Tradeshows Vet Show – London Vet Show: Overview: The London Vet Show is one of the largest veterinary events in Europe, bringing together professionals, exhibitors, and speakers from various veterinary fields. It covers a wide range of topics, including clinical practice, business management, and the latest advancements in veterinary medicine. Dates: 14-15 November, 2024 Location: London, UK. European Veterinary Emergency & Critical Care Conference (EVECC): Overview: The European Veterinary Conference is a prestigious event that gathers veterinarians, researchers, and industry experts. It provides a platform for sharing knowledge on veterinary emergency and critical care. Dates: 30 May – 1 June, 2024 Location: Gothenburg, Sweden FECAVA EuroCongress: Overview: The Federation of European Companion Animal Veterinary Associations (FECAVA) EuroCongress is a major event for companion animal veterinarians. It provides a platform for learning about new developments, networking, and sharing experiences in small animal veterinary medicine. Dates: 12-14 September, 2024 Location: Athens, Greece SCIVAC: Overview: SCIVAC, the Società Culturale Italiana Veterinari per Animali da Compagnia, is a leading trade show for the veterinary industry in Italy. Held annually in Cremona, Italy, SCIVAC attracts thousands of veterinarians, breeders, pet owners, and other industry professionals from around the world. Dates: 24-25 May, 2024 Location: Rimini, Italy AFVAC: Overview: AFVAC stands for Association Française des Vétérinaires pour Animaux de Compagnie, which translates to the French Association of Veterinarians for Companion Animals. AFVAC Le Congrès is a prominent event that brings together veterinarians, breeders, pet owners, and other industry professionals from around the globe to connect, learn, and explore the latest advancements in veterinary medicine. Dates: 21-23 November, 2024 Location: Lyon, France As we anticipate the impactful discussions and breakthroughs that these events will bring, we extend our best wishes to all those dedicated to advancing veterinary healthcare in Europe. How GrowthMedics Can Help The European veterinary healthcare market offers significant opportunities, but navigating its complexities requires expertise. At GrowthMedics, we: Identify opportunities in the veterinary pharmaceuticals market and other key segments. Connect you with leading veterinary OEMs and distribution networks. Tailor strategies for success in diverse markets across Europe. Click here to know more about our veterinary industry expertise and how we helped a US based veterinary device manufacturer in their